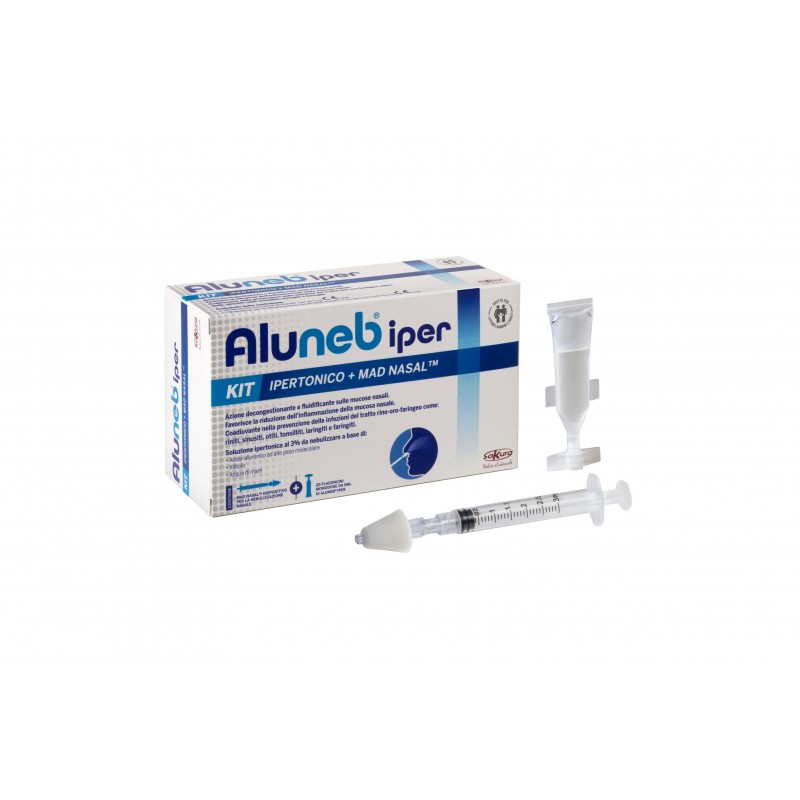

Aluneb iper kit 20 flc + MAD Nasal™
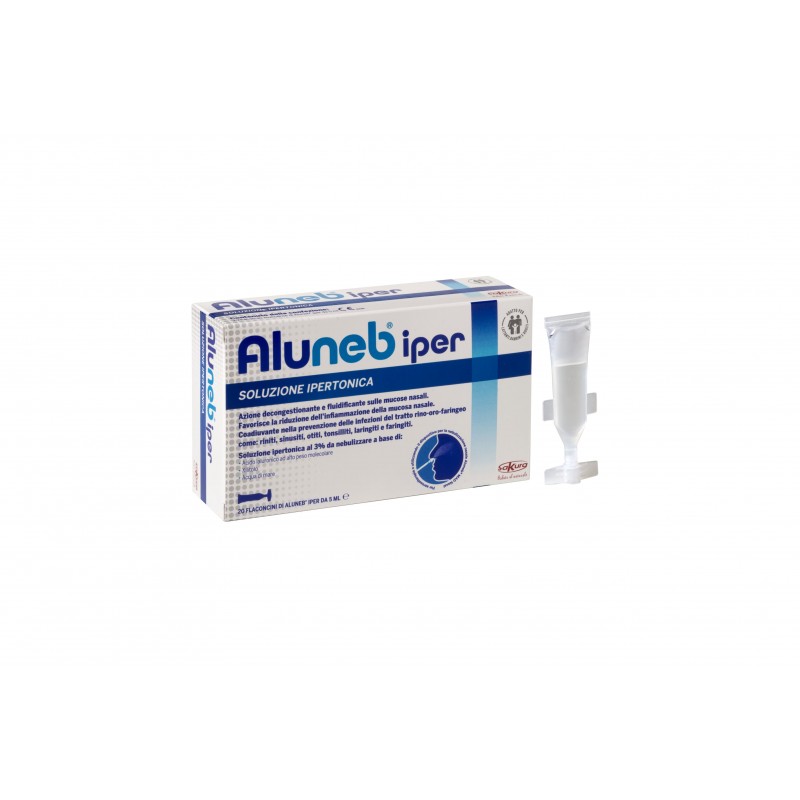
Aluneb® iper is a 3% hypertonic saline solution to be nebulized based on high molecular weight hyaluronic acid and Xylitol with decongestant and fluidifying action on the nasal mucosa. Particularly suitable for cleaning the nasal cavities especially in case of colds, rhinitis, allergic rhinitis, sinusitis, ear infections and all states of congestion of the nasal mucosa.
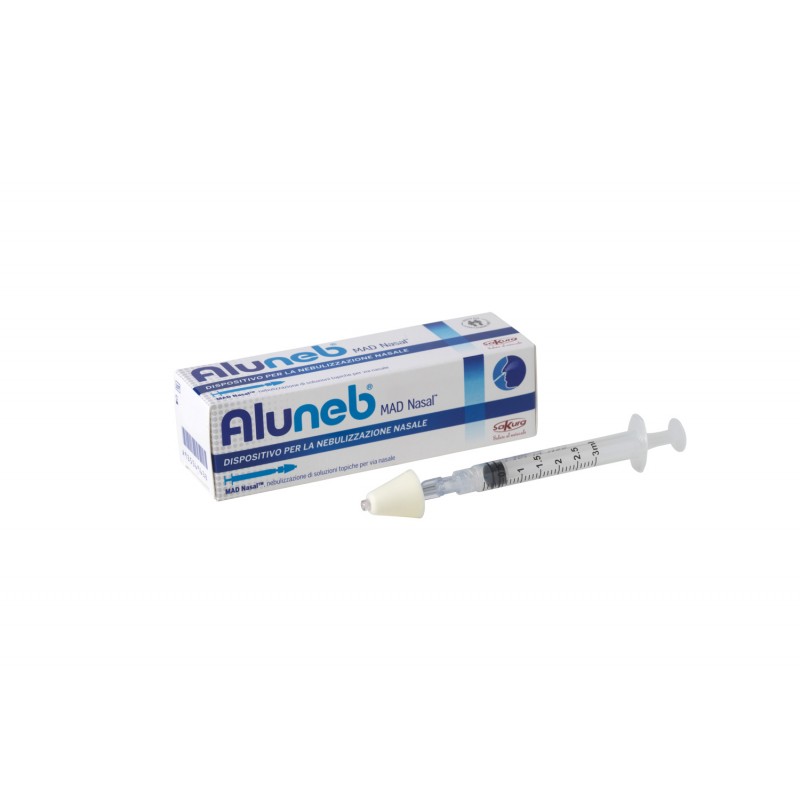
MAD Nasal ™ is a needle-free medical device for the nebulization of topical solutions via the nose. It allows the rapid administration of the Aluneb® solution through nebulisation in particles with a size of 30-100 microns in diameter ideal for upper airway therapy.
For children and adults use
Preservative-free
PACKAGE
- 20 vials of Aluneb® iper 5 ml
- MAD Nasal™ Intranasal Mucosal Nebulizer Device, 1 syringe Luer Lock 3 ml
Medical device
The legislation on the advertising of medical devices (Legislative Decree no. 46/97, as amended by Legislative Decree no. 37/10) provides that access to medical records for medical devices is restricted to authorized personnel. For more information about our products please consult your doctor or pharmacist of confidence.
Sodium chloride, Sodium hyaluronate, Xylitol, monobasic potassium phosphate, dibasic potassium phosphate, sea water and water.
Aluneb® iper
Aluneb® iper solution can be used for nebulization in aerosol therapy, nasal douche or using the Aluneb® MAD Nasal ™ nasal nebulization device.
MAD Nasal ™
Instructions
1- Disconnect the MAD Nasal™ device by unscrewing it from the supplied syringe
2 - Open the single-dose vial by rotating the wings
3 - Connect the vial to the syringe and aspirate half of the solution contained in the vial
4 - Fix the MAD Nasal ™ device by screwing it to the syringe
5 - Insert the tip of the MAD Nasal ™ device into a nostril. Keep your body in an upright position. Push the syringe plunger vigorously to atomize the solution
6 - Repeat the operations, from point 3 to point 5, also for the other nostril
Aluneb® iper
Keep out of the reach and sight of children. Avoid contact with eyes. Do not swallow. Keep the package tightly closed in a dry place, away from light. Do not use the product if the package is not intact. Do not use the product after the expiration date. The expiry date is indicated on the package and on the individual single-dose vials. Do not exceed the recommended dose without your doctor's advice. The use, especially if prolonged, of all products for topical use could give rise to sensitization phenomena. Where this occurs, stop treatment and consult your doctor in order to establish appropriate therapy. If you experience any undesirable effects described and not in this package leaflet, please inform your doctor or pharmacist. In children under one year of age and in case of bronchial asthma, administer on medical advice. In the event of concomitant use of other products, check the interactions. After use discard the vials even if residual of the product: any reuse could cause patient infection. It must not be used in case of known hypersensitivity or allergy to one of the components or other related chemicals.
MAD Nasal™
Do not use MAD NasalTM after the expiry date stated on the package. Do not use MAD NasalTM if the package is opened or damaged. Store MAD NasalTM away from hot surfaces and protect from sunlight. Store in a dry place between + 5 ° C and + 25 ° C. Do not freeze. Before each use check that the MAD NasalTM device is not damaged; if it is damaged, do not use it for nebulization. For reuse, clean following the instructions given in the paragraph "Cleaning MAD NasalTM". The device must be used for no more than 100 sprays and in any case for no more than one month. The use of MAD NasalTM by children and non self-sufficient persons must be under the supervision of an adult who has read these instructions for use. You should consult your physician before using MAD NasalTM. Keep out of the reach and sight of children. Multiple patient use of products intended for single patient use may result in degraded performance or impaired functionality of the products and may cause exposure to pathogens. There are no validated cleaning and sterilization methods available for these products, nor are there any instructions for reprocessing to original specifications.